About Uterine Fibroids
- Basic anatomy and physiology of the uterus
- What are fibroids?
- How do I know if I have fibroids?
- Surgical treatment for fibroids
- Non-surgical treatment for fibroids
- Theories of Fibroid formation
- Fibroid-like conditions: Adenomyosis
- Fibroid-like conditions: Endometrial polyps
- Medical treatments for fibroids
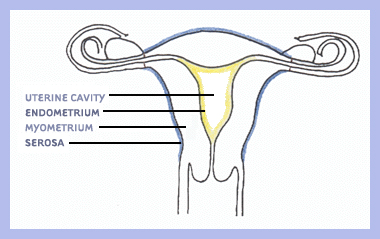
Basic Anatomy and Physiology of the Uterus
The human uterus is a pear-shaped organ composed of two distinct anatomic regions: the cervix and the corpus.
The corpus is further divided into the lower uterine segment and the fundus. The cervix is a narrow cylindrical passage which connects at its lower end with the vagina. At its upper end, the cervix widens to form the lower uterine segment (isthmus); the lower uterine segment in turn widens into the uterine fundus.
The corpus is the body of the uterus which grows during pregnancy to carry a fetus.
Extending from the top of the uterus on either side are the fallopian tubes (oviducts); these tubes are continuous with the uterine cavity and allow the passage of an ova (egg) from the ovaries to the uterus where the egg may implant if fertilized.
The thick wall of the uterus is formed of three layers: endometrium, myometrium, and serosa. The endometrium (uterine mucosa) is the innermost layer that lines the cavity of the uterus.Throughout the menstrual cycle, the endometrium grows progressively thicker with a rich blood supply to prepare the uterus for potential implantation of an embryo. In the absence of implantation, a portion of this layer is shed during menstruation.
The myometrium is the middle and thickest layer of the uterus and is composed of smooth (involuntary) muscle. The myometrium contracts during menstruation to help expel the sloughed endometrial lining and during childbirth to propel the fetus out of the uterus. The outermost layer, or serosa, is a thin fibrous layer contiguous with extrauterine connective tissue structures such as ligaments that give mechanical support to the uterus within the pelvic cavity. Non-pregnant uterine size varies with age and number of pregnancies, but is approximately three and a half inches long and weighs about one sixth of a pound.
What are Fibroids?
Uterine leiomyomas, commonly known as fibroids, are well-circumscribed, non-cancerous tumors arising from the myometrium (smooth muscle layer) of the uterus. In addition to smooth muscle, leiomyomas are also composed of extracellular matrix (i.e., collagen, proteoglycan, fibronectin). Other names for these tumors include fibromyomas, fibromas, myofibromas, and myomas.
Leiomyomas are the most common solid pelvic tumor in women, causing symptoms in approximately 25% of reproductive age women. However, with careful pathologic inspection of the uterus, the overall prevalence of leiomyomas increases to over 70%, because leiomyomas can be present but not symptomatic in many women. The average affected uterus has six to seven fibroids.
Leiomyomas are usually detected in women in their 30's and 40's and will shrink after menopause in the absence of post-menopausal estrogen replacement therapy. They are two to five times more prevalent in black women than white women. Risk for developing leiomyomas is also higher in women who are heavy for their height and is lower in women who are smokers and in women who have given birth. Although the high estrogen levels in oral contraceptive pills has led some clinicians to advise women with leiomyomas to avoid using them, there is good epidemiologic evidence to suggest that oral contraceptive use decreases the risk of leiomyomas.
Leiomyomas are classified by their location in the uterus. Subserosal leiomyomas are located just under the uterine serosa and may be pedunculated (attached to the corpus by a narrow stalk) or sessile (broad-based). Intramural leiomyomas are found predominantly within the thick myometrium but may distort the uterine cavity or cause an irregular external uterine contour. Submucous leiomyomas are located just under the uterine mucosa (endometrium) and, like subserosal leiomyomas, may be either pedunculated or sessile. Tumors in subserosal and intramural locations comprise the majority (95%) of all leiomyomas; submucous leiomyomas make up the remaining 5%.
Although this classification scheme is widely used by clinicians, it suffers from the limitation that few leiomyomas are actually a single "pure" type. Most leiomyomas span more than one anatomic location and, therefore, are hybrids (e.g., a predominantly intramural leiomyoma with a submucous component). Other types of leiomyomas include "parasitic" myomas, which receive their blood supply from structures other than the uterus (e.g., the omentum), and seedling myomas, which have a diameter of less than or equal to four millimeters.
Transformation of uterine leiomyomas (benign) to uterine leiomyosarcomas (malignant smooth muscle tumors of the uterus) is extremely rare, and, in fact, many researchers and clinicians believe this type of transformation never occurs. However, without pathologic examination of the uterus, this determination is not possible. Uterine leiomyosarcomas are found in approximately 0.1% of women with leiomyomas and are reported to be more frequently associated with large or rapidly growing fibroids. Therefore, surgical intervention may be undertaken in women with these types of tumors to rule out leiomyosarcoma, a rare but medically important lesion.
How do I know if I have fibroids?
Symptoms
Research indicates that between 20% and 50% of women have fibroid-related symptoms. The two most common symptoms of fibroids (also called leiomyomas) are abnormal uterine bleeding and pelvic pressure.
The most common bleeding abnormality is menorrhagia (prolonged and/or profuse uterine bleeding, also called hypermenorrhea). Normal menstrual periods typically last four to five days, whereas women with fibroids often have periods lasting longer than seven days. Women with fibroids also can have such heavy bleeding that they need to change sanitary protection frequently (perhaps every hour) or hesitate to participate in their normal activities for fear of socially embarrassing bleeding. Bleeding between periods is not usually associated with fibroids and should always be investigated by a physician. Although abnormal bleeding can occur with any of the three classes of fibroids, women with submucous fibroids seem particularly prone to this complication.
Pelvic pressure results from an increase in size of the uterus or from a particular fibroid. Most women with leiomyomas have an enlarged uterus; in fact, doctors describe the size of a uterus with fibroids as they would a pregnant uterus, for example, as a 12 week-size fibroid uterus. It is not unusual for a uterus with leiomyomas to reach the size of a four to five month pregnancy. In addition to vague feelings of pressure because a fibroid uterus is usually irregularly shaped (having many lumps and bumps), women can experience pressure on specific adjacent pelvic structures including the bowel and/or bladder. Pressure on these structures can result in difficulty with bowel movements and constipation or urinary frequency and incontinence. Rarely, fibroids can press on the ureters (which carry urine from the kidneys to the bladder) which can lead to kidney dysfunction.
Leiomyomas are also associated with a range of reproductive dysfunction including recurrent miscarriage, infertility, premature labor, fetal malpresentations, and complications of labor. Although few studies exist regarding fibroid-related reproductive dysfunction, the prevailing clinical perspective is that these complications most often occur when fibroids physically distort the uterine cavity. Therefore, women with large or symptomatic fibroids may choose to undergo assessment of the uterine cavity (such as by hysterosalpingograpy or by hysteroscopy, see below) before attempting pregnancy. If fibroids are detected on the inside of the uterus (termed submucous fibroids) and distort the uterine lining, they are a significant cause of reproductive problems and should be removed. It is less clear whether fibroids in the wall of the uterus cause reproductive problems. Generally, if the uterus is small, fibroids do not need to be removed in women contemplating or attempting pregnancy.
Diagnosis
The diagnosis of leiomyomas is usually easily determined by bimanual pelvic examination. During this routine office exam, the clinician evaluates the size and shape of the uterus and surrounding pelvic structures by inserting two fingers of one hand into the vagina while palpating the patient's abdomen above the pubic bone with the other hand. During this exam, a uterus with fibroids often feels enlarged and/or irregular and may be felt abdominally above the pubic bone. In contrast, a non-pregnant uterus without fibroids is not palpable above the pubic bone.
In addition, imaging studies such as ultrasonography, MRI (magnetic resonance imagery), and CT (computed tomography) may be useful in confirming the diagnosis. Currently, ultrasonography is the most common method of confirming the diagnosis of leiomyomas, but MRI may prove to be the most useful method because it can often distinguish leiomyomas from other intramural lesions.
In patients experiencing menorrhagia (profuse and/or prolonged menstrual flow) or recurrent pregnancy losses, assessment of the uterine cavity is important because the presence of a submucous fibroid can be missed on traditional ultrasound.
Hysterosalpingography, sonohysterography, and hysteroscopy can all supply this information and aid in a more definitive diagnosis of fibroids. More invasive procedures such as laparoscopy can also aid in definitive diagnosis. Hysterosalpingography and sonohysterosgraphy use X-ray pictures and ultrasound pictures, respectively, to visualize the uterine cavity after a specific dye is injected into the uterus. Hysteroscopy allows direct visualization of the uterine cavity by inserting a small camera on the end of a long tube (hysteroscope) directly into the uterus through the vagina and cervix. Laparoscopy, on the other hand, allows direct visualization of the outside of the uterus and the surrounding pelvic structures by introducing a small camera on the end of a tube (laparoscope) directly into the abdominal cavity.
Surgical treatments for fibroids
Hysterectomy
In general, fibroids only need to be treated if they are causing symptoms. The primary treatment for patients with large or symptomatic fibroids is surgery. Hysterectomy (surgical removal of the entire uterus) is the most frequent operative technique used to treat this disorder. In fact, fibroids are the most common indication for hysterectomy, accounting for approximately one third of hysterectomies, or about 200,000 procedures annually, in the United States.
There are a variety of types of hysterectomy including abdominal hysterectomy, vaginal hysterectomy, supracervical hysterectomy, and laparoscopically-assisted vaginal hysterectomy. The type of hysterectomy chosen depends on the size of the uterus, the woman's medical history, and the skills of her surgeon. The advantage of hysterectomy in the treatment of leiomyomas is that it provides a true "cure" for fibroids, but is only an option for women who are not planning future pregnancies.
Myomectomy
When women wish to preserve childbearing potential, a myomectomy may be performed. Unlike hysterectomy in which the entire uterus is removed, myomectomy is a surgical procedure in which individual fibroid(s) are removed. Approximately 18,000 myomectomies are performed yearly in the United States. Most myomectomies are performed through an abdominal incision, although certain submucous fibroids can be removed through the vagina without an abdominal incision during a procedure called hysterosopic myomectomy which involves a special instrument called a hysteroscope. This technique is primarily useful for women with bleeding or pregnancy-related problems as there is usually little change in the size of the uterus with this approach. Certain subserosal fibroids may be removed abdominally during a procedure called laparoscopic myomectomy which involves a different instrument called a laparoscope. In general, myomectomy diminishes menorrhagia (prolonged and/or profuse menstrual flow) in roughly 80% of patients presenting with this symptom. Unfortunately, there is a significant risk of recurrence of fibroids after myomectomy; in some studies up to 10% of women who underwent an initial myomectomy required a second major operative procedure. In addition, a quarter to a half of women who underwent myomectomies had evidence by ultrasound of recurrence of their fibroids within one to ten years.
Fibroids that are in the wall or outer lining of the uterus (intramural and subserosal fibroids) can be removed either through an abdominal myomectomy or a laparoscopic approach. An abdominal myomectomy involves making an incision on the abdomen in order to access the uterus and then surgically remove the fibroids. Either a Pfannenstiel or vertical skin incision can be used to perform an abdominal myomectomy. A Pfannenstiel skin incision is a horizontal incision that made just above the pubic bone, and vertical skin incision is an up and down incision that extends from the pubic bone to the umbilicus (belly button) or possibly above the umbilicus. The type of incision made depends on the size of the uterus and fibroids. Both a laparoscopic myomectomy and robotic myomectomy are minimally invasive procedures that similarly remove intramural and subserosal fibroids, but because they are minimally invasive, allow for a quicker recovery and less scar tissue formation as compared to a traditional abdominal myomectomy. These surgical approaches use 3-5 small abdominal skin incisions that are 5-12 mm to perform the same surgery. A laparoscope, or thin long camera, is placed through an incision in the umbilicus or above the umbilicus, depending on the size of the uterus and uterine fibroids. Long instruments are then inserted through the remaining small incisions to perform the removal of the fibroids and then to repair of the uterus. Robotic myomectomies are performed using specialized equipment that allows for free range of motion and a three-dimensional high definition view of the surgical anatomy. See a live-recorded video of robotic myomectomy.
There are also several innovative techniques being studied as possible surgical treatment for fibroid-related bleeding. Myolysis involves delivering electric current via needles to a fibroid at the time of laparoscopy. Cryomyolysis involves using a freezing probe in a similar manner. Uterine artery embolization is a radiological alternative to surgery that involves placing a catheter into an artery in the leg and guiding the catheter via x-ray pictures to the arteries of the uterus. Once there, the catheter is used to deliver agents that block off these major blood vessels. While all of these treatments may prove to be effective treatments for fibroids, compared to more traditional options, the number of patients treated by these methods have been small, the follow-up relatively short term, and the safety of these procedures in women desiring pregnancy has not been demonstrated.
Women need to talk to their physicians to determine the best surgical approach for them based on their medical history, size of their uterus and number of uterine fibroids. In general, myomectomy diminishes menorrhagia (prolonged and/or profuse menstrual flow) in roughly 80% of patients presenting with this symptom. Unfortunately, there is a significant risk of recurrence of fibroids after myomectomy; in some studies up to 10% of women who underwent an initial myomectomy required a second major operative procedure. In addition, a quarter to a half of women who underwent myomectomies had evidence by ultrasound of recurrence of their fibroids within one to ten years. Find out more information about robotic myomectomy at Brigham and Women’s hospital.
There are also several innovative techniques being studied as possible surgical treatment for fibroid-related bleeding. Myolysis involves delivering electric current via needles to a fibroid at the time of laparoscopy. Cryomyolysis involves using a freezing probe in a similar manner.
Non-surgical treatment for fibroids
Focused Ultrasound Treatment
MRI-guided focused ultrasound surgery (FUS) is a noninvasive treatment option for uterine fibroids that preserves your uterus.
This procedure is performed inside a specially crafted MRI scanner that allows your doctors to visualize your anatomy, and then locate and destroy (ablate) fibroids inside your uterus without making an incision. Focused high-frequency, high-energy sound waves are used to target the proteins in fibroids, until they are denatured and cell death occurs, thus destroying the fibroids. Concurrent MRI allows precise targeting of tissue and monitoring of therapy by assessing the temperature of treated tissue. A single treatment session is done in an on- and off fashion, sometimes spanning several hours. The advantages of this procedure are a very low morbidity and a very rapid recovery, with return to normal activity in 1 day, but its long-term effectiveness is not yet known. Presently, the procedure is not recommended for women who desire future fertility. See a video about focused ultrasound treatment for fibroids.
If you are looking for more information, or would like to contact a physician who offers this treatment at Brigham and Women’s hospital, please contact Louise Greenberg at 617-732-5441 Toll free: 1-800-722-5441 or email lgreenberg@partners.org.
Uterine Fibroid Embolization
Uterine fibroid embolization (UFE), also known as uterine artery embolization(UAE) is a radiological alternative to surgery that involves placing a catheter through a small incision in the groin into an artery in the leg and guiding the catheter via x-ray pictures to the arteries of the uterus. Once there, the catheter is used to deliver agents that block off the blood vessels that feed the uterine fibroids. Specific embolic agents include gelatin sponges, polyvinyl alcohol particles, or tris-acryl gelatin microspheres. Total radiation exposure, following this procedure is approximately 15 rads, which is comparable to that in one to two computed tomography scans or barium enemas1 (ref). UFE is a minimally invasive technique that allows for quick recovery after the procedure. UFE does not remove uterine fibroids, but causes the fibroids to shrink about 30-50%. Women can have improvement in both their bleeding symptoms and pressure symptoms after UFE. A pelvic MRI is performed before the procedure to determine if a woman would benefit from a UFE. Women also need to have a physical examination and discuss their medical history with an interventional radiologist to determine if they are suitable candidate for a UFE procedure. Advantages over surgery include no abdominal incisions and a shorter recovery time. Complications may occur if the blood supply to your ovaries or other organs is compromised. The safety of these procedures in women desiring pregnancy has not been demonstrated and is generally not advised for women who want to have children in the future. Learn more about Uterine Fibroid Embolization at Brigham & Women's Hospital.
Theories of Fibroid Formation
Despite the major public health impact of leiomyomas, little is known about their cause. Until recently, the steroid hormones estrogen and progesterone were considered the most important regulators of leiomyoma growth. There is abundant evidence that estrogen promotes fibroid growth including the clinical observations that fibroids grow in the presence of high levels of estrogen, such as during the reproductive years, and that they regress in the presence of low levels of estrogen, such as following menopause or during gonadotropin releasing hormone (GnRH) agonist therapy. Furthermore, fibroids have higher estrogen concentrations, bind more estrogen, have more estrogen receptors, and convert estradiol (a more active form of estrogen) to estrone (a less active form of estrogen) more slowly than normal myometrium.
Progesterone is also thought to play a role in fibroid growth. More specifically, clinical studies suggest progesterone facilitates the growth of fibroids. For example, fibroid size increases during treatment with synthetic progesterones. Combination GnRH agonist and progesterone therapy has been shown to have no effect on uterine volume, in contrast to GnRH agonist therapy alone which has been shown to reduce uterine volume. The observation that fibroids regress with the antiprogesterone agent, RU-486, further supports the role of progesterone as a promoter of fibroid growth. Histologically, fibroids from patients treated with progesterone show more cellular growth than those from patients without progesterone therapy. Biochemically, fibroids have higher progesterone receptor concentrations than normal myometrium. Together, these data suggest that progesterone also enhances fibroid growth.
Other hormones such as growth hormone (GH) and prolactin (PRL) are also thought to promote fibroid growth, but their role is even less well defined.
More recently, growth factors, which are small proteins that affect cell growth, have been shown to mediate the growth-promoting effects of estrogen and to play an important role in the development of fibroid tumors. Potentially important factors in fibroid growth include transforming growth factor-beta, basic fibroblast growth factor, epidermal growth factor, insulin-like growth factor, and platelet-derived growth factor. (For more information about this, please see the article, Leiomyoma-related bleeding: A classic hypothesis updated for the molecular era, on the "Publications" page.)
Overall, estrogen, progesterone, and growth factors likely promote tumor growth, but only after the initiation of tumor formation. This initiating event remains unknown, although recent evidence suggests there is a strong inherited component to fibroid development. Indirect evidence for this hypothesis is as follows. First, fibroids are at least twice as common in black women than in white women. Although racial differences in socioeconomic status and access to health care, as well as racial differences in known risk factors for fibroids, may contribute to this finding, two recent studies suggest that these factors do not completely explain the discrepancy. Secondly, another study found a genetic predisposition for hysterectomy as indicated by a two fold higher twin pair correlation for hysterectomy in identical versus fraternal twins. Thirdly, there exists a rare heritable form of uterine fibroids in association with fibroids of the skin called Reed's syndrome. Finally, a recent Russian studies suggest that women with a family history of fibroids are twice as likely to develop fibroids than women with no family history. Unfortunately, few scientific studies directly examine the genetic component of fibroid development.
Recently, researchers at the Center for Uterine Fibroids have identified mutations in two genes, HMGI(C) and HMGI(Y), that appear to be important in the development of some fibroids. (For more information about the genetics of fibroids, please see articles published about these genes on the "Publications" page.) Normally, these genes code for proteins that help control cell growth by indirectly regulating DNA transcription.
However, mutations in these genes are probably secondary changes in already genetically susceptible cells. Therefore, it is likely that other gene(s) crucial for fibroid development exist that have not yet been identified. To this end, the staff at the Center for Uterine Fibroids is studying families with at least one pair of siblings affected by fibroids to search for gene(s) that predispose women to fibroid development. For information about this study, including participation, please see, Finding Genes for Fibroids, on the "Current Studies" page.
Ultimately, understanding the hormones, growth factors, and gene(s) involved in the formation and growth of fibroid tumors may lead to innovative, less invasive treatment options.
Fibroid-like Conditions: Adenomyosis
Definition, prevalence and causes
Adenomyosis is a benign disease of the uterus in which components normally limited to the endometrium (the thin innermost uterine layer) are found within the myometrium (the middle muscular layer of the uterus). The exact prevalence of adenomyosis is not known because the diagnosis can be made only by microscopic examination of uterine specimens obtained during surgery or, less often, during biopsy. Some studies estimate that 20% of women have adenomyosis; however, with careful microscopic analysis of multiple myometrial samples from an individual uterine specimen, the prevalence increases to as high as 65%.
The cause of adenomyosis is also unknown. The most widely accepted theory of adenomyosis development postulates that the barrier between the endometrium and myometrium, which normally prevents invasion of endometrial glands and stroma into the myometrium, is compromised allowing invasion to occur. This process is thought to occur only in the presence of estrogen, however, little scientific evidences exists to support this hypothesis.
Adenomyosis most commonly affects women between the ages of 40 and 50 years and is associated with a past history of childbirth. Approximately 80% of women with this disorder have given birth. However, the incidence of adenomyosis does not correlate with increasing number of pregnancies.
Adenomyosis is also associated with other uterine disorders. More than 80% of women with adenomyosis have another pathologic process in the uterus; 50% of patients have associated fibroids (benign smooth muscle tumors of the uterus), approximately 11% have endometriosis (endometrial tissue outside of the uterus, most commonly in the ovaries), and 7% have endometrial polyps (benign outgrowths of endometrial tissue). The symptoms of these associated conditions often obscure the diagnosis of adenomyosis.
Symptoms and diagnosis
A typical uterus with adenomyosis is boggy and uniformly enlarged. Approximately 80% of uteri with adenomyosis weigh more than 80 grams (a "normal" uterus weighs approximately 50 grams), but it is unusual for a uterus in which adenomyosis is the only pathologic process to exceed 200 grams.
Symptoms of adenomyosis include abnormal uterine bleeding and pelvic pain. Approximately 60% of women with adenomyosis experience abnormal uterine bleeding which usually manifests as either hypermenorrhea (prolonged and/or profuse uterine bleeding, also called menorrhagia) or metrorragia (irregular, acyclic bleeding). Dysmenorrhea (pelvic pain during menstruation) is the second most common symptom in patients with adenomyosis, occurring in 25% of cases.
A review of the literature demonstrates that only 15% of cases of adenomyosis are correctly diagnosed before surgery. The reason for this low percentage of preoperative diagnosis is two-fold; first, many patients with adenomyosis are asymptomatic in the absence of other uterine pathology, and second, the presence of adenomyosis is often overshadowed by associated pathology (e.g., leiomyomas, endometriosis).
D&C (dilation and curettage) does not aid in diagnosis. (In this procedure, the cervix is gradually dilated to allow removal of the uterine lining.) Pelvic ultrasonography may be suggestive but is not definitive. The usefulness of other imaging studies such as MRI (magnetic resonance imaging) is currently undetermined.
Treatment
Areas of adenomyosis do not lend themselves to local surgical excision. The only definitive treatment for adenomyosis, therefore, is total hysterectomy (surgical removal of the entire uterus). Synthetic steroid hormones such as progestins are not helpful and may actually increase the level of pelvic pain in some patients. GnRH (gonadotropin releasing hormone) agonists have been used in a few cases, resulting in a transient decrease in uterine size, in amenorrhea (cessation of menstrual cycling), and even in the ability to conceive. Unfortunately, regrowth of the adenomyosis and recurrence of symptoms are usually documented within six months of cessation of therapy.
Fibroid-like Conditions: Endometrial Polyps
Definition and prevalence
Endometrial polyps are localized overgrowths of the endometrium (innermost uterine layer) that project into the uterine cavity. Such polyps may be sessile (broad-based) or pedunculated (on a narrow stalk) and rarely include areas of neoplastic (benign or malignant) growth. Specifically, adenomatous hyperplasia (benign growth of the endometrium) and endometrial adenocarcinomas (malignant tumors of the glandular component of the endometrium), have been reported in only 0.6% of cases of endometrial polyps.
The prevalence of polyps is estimated to be 10% to 24% of women undergoing hysterectomy (surgical removal of the uterus) or localized endometrial biopsy. Endometrial polyps are rare among women younger than 20 years of age. The incidence of these polyps rises steadily with increasing age, peaks in the fifth decade of life, and gradually declines after menopause.
Symptoms and Diagnosis
The most frequent symptom of women with endometrial polyps is metrorrhagia (irregular, acyclic uterine bleeding), which is reported in 50% of symptomatic cases. Post-menstrual spotting is also common. Less frequent symptoms include hypermenorrhea (prolonged and/or profuse uterine bleeding, also called menorrhagia), post-menopausal bleeding, and breakthrough bleeding during hormonal therapy. Overall, endometrial polyps account for 25% of abnormal bleeding in both premenopausal and postmenopausal women.
Endometrial polyps are often diagnosed by microscopic examination of a specimen obtained after endometrial biopsy or after D&C (dilation and curettage); in this latter procedure, the cervix is gradually dilated to allow removal of the uterine lining. As with submucous fibroids, the diagnosis of polyps can be missed on physical exam if the uterus is not distended. Therefore, these lesions are being increasingly diagnosed by techniques such as ultrasound and hysteroscopy. During hysteroscopy, the uterine cavity is visualized by introducing a small camera on the end of a tube (hysteroscope) directly into the uterus through the vagina and cervix. Hysteroscopy with directed biopsy is particularly helpful in the diagnosis of small polyps within the uterine cavity. Hysterography, a technique using X-rays to take pictures of the uterine cavity, is rarely helpful when polyps are small but may yield suggestive findings (e.g., a smooth space-occupying lesion) when the polyp is large.
Treatment
The majority of cases of endometrial polyps are cured by thorough curettage. This technique, which involves removing the endometrial lining of the uterus, is especially successful in the post-menopausal age group. However, removal of polyps or other structural abnormalities may be missed by blind curettage, therefore, hysteroscopic-guided curettage is often useful.
Medical Treatments for Fibroids
Medicines can help control fibroid-related symptoms. The most effective medications for the treatment of fibroids are gonadotropin releasing hormone agonists (GnRHa), (including Lupron, Synarel, Zoladex). GnRH agonists induce a low-estrogen (menopause-like) state. Because fibroids are dependent on estrogen for their development and growth, induction of a low estrogen state causes reduction of tumor and uterus mass, resolving pressure symptoms. (Specifically, uterine volume has been shown to decrease approximately 50% after three months of GnRH agonist therapy.) In addition to decreasing the size of the uterus, treatment with GnRH agonists also stops menstrual flow (amenorrhea), allowing women with bleeding-induced anemia to significantly increase their iron stores. Unfortunately, cessation of GnRH agonist treatment is followed by a rapid regrowth of the fibroids and of the uterus to pre-treatment volume. Additionally, because bone also requires estrogen, long term use of GnRH agonists can significantly decrease bone density and can lead to bone loss or osteoporosis. Currently, therefore, use of GnRH agonists alone for treatment of fibroids is usually limited to a short one to three month preoperative course to shrink the uterus to facilitate a surgical procedure or to induce amenorrhea to improve hematologic condition before surgery.
The combination of GnRH agonists and low doses of the steroid hormones estrogen and progesterone (i.e., "add-back" regimens) has been employed in some clinical trials in an attempt to safely extend the maximum duration of GnRH agonist therapy without sacrificing efficacy. These regimens have been studied for use of up to two years. Preliminary data suggest they may be safe and effective if the hormone dose is low (equivalent to menopausal replacement doses versus high dose birth control pills) and if the GnRH-agonist is given alone first, allowing the uterus to shrink before the hormones are added. This approach appears to maintain uterine shrinkage and/or control of bleeding while supporting other tissues such as bone and minimizing side effects such as hot flashes that accompany the low estrogen levels induced by GnRH-agonist therapy alone.
Several innovative options are under investigation as possible future medical treatments for uterine leiomyomas. There have been several small studies examining the use of GnRH antagonists in leiomyomas. The primary advantage of antagonists over more widely used GnRH agonists is that antagonists have a faster onset of action. However for long-term therapy, antagonists appear to have no advantage. The progesterone antagonist, mifepristone (RU 486), has also been shown in small studies to induce uterine shrinkage and stop menstrual periods in women with fibroids. However, this agent is not currently available in the United States. Studies of the antifibrotic drug, pirfenidone, are also underway to determine if this agent is useful in the treatment of fibroids.
Other medical therapies including androgenic agents (e.g., danazol, gestrinone), progestins (e.g., medroxyprogesterone acetate, depomedroxyprogesterone acetate, norethindrone), and oral contraceptive pills have also been used to control menorrhagia (prolonged and/or profuse blood flow) in women with leiomyomas, presumably by diminishing the endometrium (endometrial atrophy). However, these medications do not consistently decrease uterus or fibroid volume and are often ineffective in controlling menorrhagia.