As published in Molecular Human Reproduction
vol. 3 no. 8 pp. 685-691, 1997
Departments of Obstetrics, Gynecology and Reproductive Biology and Pathology, Brigham and Women's Hospital, Harvard Medical School, Boston, Massachusetts, USA
1 To whom correspondence should be addressed at: Laboratory for Human Reproduction and Reproductive Biology, Room 449, Boston Lying-In, 221 Longwood Avenue, Boston, Massachusetts 02115, USA
Basic fibroblast growth factor (bFGF) is a
regulator of angiogenesis which is overexpressed in leiomyomas compared to
matched myometrium. To understand the physiological significance of this finding
we characterized the expression of the type 1 receptor for this ligand (FGFR1).
Utilizing RT-PCR we identified the complete and alternatively spliced
transmembrane forms and two secreted forms of the FGFR1 in endometrium,
myometrium and leiomyomas from all patients. This is the first report of
secreted forms in uterine tissue. Proteins consistent with each of these
isoforms were identified by western blot analysis in all three tissues.
Menstrual cycle specific regulation of FGFR1 protein was seen in the endometrial
stroma of normal women but not in women with leiomyomas and abnormal uterine
bleeding utilizing immunohistochemistry. Stromal FGFR1 expression is suppressed
in the early luteal phase in normal women, but not in women with leiomyoma-related
bleeding. These findings support the role of the bFGF ligand-receptor system in
the pathogenesis of leiomyoma-related bleeding and may have implications for
fertility and contraception since the differential FGFR1 expression occurs in
the peri-implantation period of the early luteal phase.
Key words: angiogenesis/fibroblast growth factor/leiomyoma/ uterine
bleeding
Introduction
Despite the central role that the endometrial vasculature must play in any gynecologic bleeding disorder, few studies have evaluated endometrial microvasculature and angiogenesis in women with abnormal uterine bleeding. Angiogenesis is a complex process that occurs in many pathological conditions. As a physiological process, angiogenesis occurs primarily in the female reproductive tract, in particular, vascularization of the endometrium during the menstrual cycle and development of the corpus luteum.
Ovarian steroids are the classic regulators of endometrial development, but recent evidence indicates that growth factors, either alone or in conjunction with steroids play an important role in endometrial growth (Irwin et al., 1991). Altered expression of these factors may affect endometrial development by causing local abnormalities in vascular structure and/or function and these abnormalities may be important in the pathogenesis of abnormal bleeding.
Basic fibroblast growth factor (bFGF) is an angiogenic growth factor that is highly mitogenic for capillary endothelial cells in vitro and can induce angiogenesis in vivo (Folkman and Klagbrun, 1987). It is present in the endometrium, myometrium and corpus luteum of women throughout the normal menstrual cycle (Ferriani et al., 1993; Rusnati et al., 1990; Salat-Baroux et al., 1994). In addition, bFGF has been shown to be stored in extracellular matrix (ECM) and can initiate remodeling of ECM, an important step in angiogenesis (Gospodarowicz, 1983). Uterine leiomyomas are benign smooth-muscle tumours that often cause abnormal bleeding, although the pathogenesis of this process is poorly understood. Leiomyomas are characterized by large amounts of ECM, and contain abundant amounts of bFGF in this ECM (Mangrulkar et al., 1995). Leiomyomas may therefore serve as a reservoir for bFGF and impact endometrial vasculature through a paracrine or local endocrine effect.
Although the expression of bFGF in the uterus has been described, to fully understand its function requires knowledge of its receptors in endometrium, myometrium and leiomyomas throughout the menstrual cycle. The gene for bFGF receptor type 1 (FGFR1) has been cloned and is known to generate multiple isoforms by alternative splicing of mRNA (Johnson et al., 1991). The complete form of the receptor is composed of three extracellular immunoglobulin-like domains, a transmembrane region, and an intracellular portion exhibiting intrinsic tyrosine kinase activity (Lee et al., 1989). An alternatively spliced transmembrane variant has been characterized that has only two immunoglobulin-like domains (Lee et al., 1989). A third isoform generated by alternative splicing encodes for a secreted form of the receptor that lacks the transmembrane and intracytoplasmic domains (Johnson et al., 1991). The functional significance of the secreted form is unknown, but it may act as a binding protein for bFGF and thus may prevent the binding between bFGF and the membrane-bound receptor. Using women with leiomyomas as a model system, we thus examined differences in receptor expression in women with and without abnormal bleeding to gain insight into the pathogenesis of abnormal uterine bleeding.
Materials and Methods
Preparation of human tissue
Samples of myometrium, endometrium and leiomyomas from the
same uterus were obtained under an approved human subjects protocol at the
Brigham and Women's Hospital. Tissue was obtained from fourteen premenopausal
women undergoing hysterectomy for leiomyoma-related bleeding. All preoperatively
had complaints of menorrhagia and documented anemia. Pathologic examination of
the uterus confirmed leiomyomas with mean uterine weight of 496 ± 431 gm and no
other pathologic reasons for bleeding. Normal control endometrial biopsy
specimens were obtained from 14 premenopausal women who were undergoing
bilateral tubal ligation who had no known uterine pathology and no evidence of
abnormal uterine bleeding. Histologic dating of the endometrium was performed
for all samples utilizing classic criteria (Noyes et al., 1950). Patients with
leiomyoma-related bleeding were comparable to control patients in terms of age
(45.8 ± 3.2 vs. 37.3 ± 6.5), gravidity (2.9 ± 2.1 vs. 3.3 ± 0.7) and parity
(2.6 ± 1.7 vs. 2.7 ± 0.8). No patient in either group received any hormonal
medication for at least two months before hysterectomy or biopsy.
Tissues were obtained within 30 minutes of surgical
resection and divided into three sections: one was minced and immediately
homogenized in 4M guanidine isothiocyanate for RNA processing, one was placed in
10% formalin for histological analysis, and the third was homogenized and
processed for membrane protein extraction.
Paraffin sections were first deparaffinized and rehydrated
in phosphate buffered saline. Sections were then incubated with a mouse
monoclonal anti-FGFR1 primary antibody [Santa Cruz Biotechnology, Inc., Santa
Cruz, California] 2 mg/ml for 30 minutes at room temperature. Nonspecific mouse
IgG (Sigma, St. Louis, MO) was used as a negative control at 5ug/ml. Binding was
visualized with the Vecta-Stain kit (Vector Laboratories, Burlingame, CA) using
diaminobenzidine tetrahydrochloride (DAB) for detection with hematoxylin
counterstaining. Negative controls were examined for every slide. Staining
intensity in tissue sections was evaluated and graded (1 weak; 2 moderate; 3
strong) in a blinded fashion by two examiners and assigned a semiquantitaive
HSCORE, assessing both intensity of staining and percentage of stained cells (Lessey
et al., 1992). Endometrial glands and stroma were assessed separately.
Intra-assay and interassay variations for this technique have been previously
validated (Budwit-Novotny et al., 1986).
HSCORE data was analyzed using the Wilcoxon rank sum test
for independent nonparametric variables utilizing STATA statistical software
(Computing Resource Center, College Station, TX). Data are reported as the mean
± standard deviation.
Total cellular RNA was extracted from fresh tissue using
guanidine isothiocyanate as previously described (Chirgwin et al., 1979). cDNA
was synthesized using 1 ug of total RNA with oligo (dT) priming (Pharmacia,
Piscataway, NJ) using a standard protocol (Nowak et al., 1993; Yeh et al.,
1993).
Three different primer pairs were used to detect the three
different forms of the FGFR1. To detect the complete transmembrane form of
FGFR1, primers used were 5'-ATGTGGAGCTGGAAGTGCCTC-3' and
5'-GCTGCTGGTTACGCAAGCATA-3' (Isacchi et al., 1990; Yeh and Osanthanondh, 1993).
These primers amplify a 315-bp region corresponding to amino acids 1 to 319 (Isacchi
et al., 1990). The FGFR1 alternatively spliced transmembrane variant was
amplified with the same upstream primer and a different downstream primer
(5'-GGTGTTATCTGTTTCTTTCTC-3') corresponding to amino acids 422 to 431 (Isacchi
et al., 1990; Yeh and Osanthanondh, 1993). These primers produce expected bands
of 432 bp (complete form) and 165 bp (alternatively spliced form). The FGFR1
secreted form was amplified utilizing a different primer pair:
5'-TATGCCACCTGGAGCATCATAATGGACTCTGTGGTGCCCTCTGAC-3' and 5'-
GACTGGCCCACGAAGACTGGTGCCAT-3' with an expected amplified band of 341 bp (Johnson
et al., 1991). These primers encompasses bases 877 to 1217. Amplification was
carried out as per our standard protocol for forty cycles with the following
thermocycling conditions: 94¡C for 30 seconds, 55¡C for 2 minutes and 72¡C
for 1 minute. Umbilical vein RNA was used as a positive control and negative
controls were used in each amplification and included all reagents, except cDNA.
The amplified products were analyzed by electrophoresis on
2% agarose gels with a 100-bp DNA ladder (Pharmacia, Piscataway, NJ) as a
molecular weight standard. All products were restriction digested: restriction
digestion of DNA from the complete form of FGFR1 by AvaII produces two
fragments, 183 and 132 bp; digestion of the alternative form of FGFR1 by Hinf1
results in fragments of 130 and 35 bp; and digestion of the secreted form by
AvaII gives fragments of 122 and 219 bp. DNA sequence analysis was also
performed on all products to confirm their identities. Gel-purified PCR products
were sequenced using an ABI 373A DNA sequencer (Perkin-Elmer, Foster City CA)
using dye-primer cycle sequencing.
Membranes were isolated from surgical specimens as
described by Mukku and Stancel (Mukko and Stancel, 1985). Tissue samples were
homogenized in 1,4-piperazine diethane sulfonic acid (PIPES) buffer with
protease inhibitors leupeptin, antipan, benzamidine, chymostatin and pepstatin
to prevent protein degredation (Mukko and Stancel, 1985). The homogenate was
centrifuged twice at 800 x g for 15 minutes at 4¡C. The resulting supernatants
were combined and centrifuged at 105,000 x g for 60 minutes at 22¡C. Membrane
pellets were resuspended and samples were centrifuged at 105,000 x g for 60
minutes at 22¡C. The final membrane pellet was suspended in PIPES buffer
containing 0.15M sodium chloride. Samples were then stored at -70¡C. The
protein concentration was determined with the Coomassie Protein Assay Reagent
Kit (Pierce, Rockford, IL). The final concentration of membrane proteins ranged
from 425 mg/ml to 1.5 mg/ml.
Immunohistochemistry
Statistical analysis
RNA isolation and cDNA synthesis
Polymerase chain reaction (PCR) amplification
Analysis and sequencing of PCR products
Isolation of cellular membranes
Immunoblotting
Immunoblots were performed with slight
modification from our standard protocol (Mangrulkar et al., 1995). Membrane
proteins (30 mg) were treated with denaturing buffer at 68¡C for three minutes
and separated on an 8.0% SDS-polyacrylamide gels under nonreducing conditions.
Proteins were transferred electrophoretically to nitrocellulose membranes [0.1um
pore size (Protran, Keene, NH)] in 0.025 M Tris, 0.192 M glycine and 20%
methanol transfer buffer overnight at 40 volts in 4¡C. To prevent non-specific
protein binding, blots were pretreated in Tris-buffered saline (TBS) containing
10% nonfat dry milk at 37¡C for 60 minutes. The blots were reacted with a
polyclonal rabbit anti-FGFR1 antibody [Upstate Biotechnology, Lake Placid, NY]
diluted 1:250 in TBS containing 0.05% Tween (TBS-T) for 30 minutes at 22¡C. The
FGFR1 antibody is reported by the manufacturer to recognize the extracellular
region of chicken, rat, and human FGFR1. The polyclonal antibody was used for
immunoblotting after we found that the monoclonal FGFR1 antibody was not as
sensitive when used on the membrane extracts. Blots were washed in TBS-T and
reacted with anti-rabbit IgG alkaline phosphatase conjugate [1:1000 dilution
(Sigma, St. Louis, MO)] for 30 minutes at 22¡C. After a final wash in TBS-T,
immunoreactive proteins were visualized with NBT/BCIP (Pierce, Rockford, IL), a
modified alkaline phosphatase substrate. Rat liver was used as a negative
control and rat brain as a positive control, for FGFR1; both were received as a
gift from Dr. Virginia Rider.
Results
Immunohistochemical analysis of FGFR1 protein localization demonstrated a significant difference in stromal cell staining between normal women and women with leiomyoma related bleeding (Figures 1 and 2). In normal women, stromal cells demonstrated significant cell-associated staining in the early proliferative and late luteal phases of the cycle with suppression of staining that begins in the late proliferative phase and extends through the mid-luteal phase. In the women with abnormal uterine bleeding and leiomyomas, this suppression does not occur. A significant difference in stromal HSCORE was seen (Table I). Although there appears to be a suppression of receptor expression in women with abnormal uterine bleeding in the late luteal phase, no significant difference was seen in stromal HSCORE in either the proliferative phase or late luteal phase. Staining for endometrial glandular cells was similar for both groups with intense homogeneous staining of the cytoplasm in both proliferative and secretory phases of the menstrual cycle
.

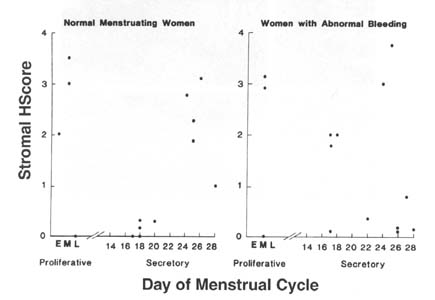
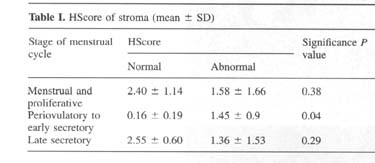
Figure 1. HSCORE for immunohistochemical
staining of endometrial stroma in women with normal menstrual cycles (left-hand
panel) and leiomyoma-related bleeding (right-hand panel). Luteal phase samples
were dated and proliferative phase samples were classified as early, mid or late
(E, M, or L). Day 28 samples were considered menstrual samples for the purpose
of analysis. A suppression of FGFR1 expression is seen in the normal women that
may begin as early as the late proliferative phase and extends through the mid-luteal
phase. This suppression is absent in most women with abnormal uterine bleeding.
In both groups, the myometrium demonstrated
homogeneous cytoplasmic staining that did not vary through the menstrual cycle
(Figure 2G, H). Overall, the intensity of the cellular staining for the receptor
was greater in the myometrium than in leiomyomas (Figure 2G, H, I, J).
Leiomyomas, however, contained large amounts of ECM, which did show some
immunoreactivity for the receptor (Figure 2I, J). Leiomyomas also exhibited
homogeneous cytoplasmic staining that did not vary through the menstrual cycle.
Matched negative controls were consistently immunonegative.
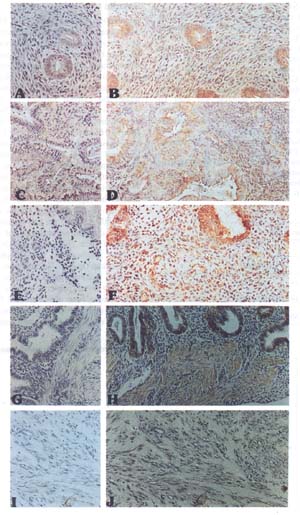 Figure 2. Immunohistochemical localization of
fibroblast grwoth factor receptor type 1 (FGFR1) in human uterine tissues
throughout the menstrual cycle. Panels A, C, E, G and I are negative controls
and were treated with nonspecific (Ig)G at a concentration of 5 ug/ml. Panels B,
D, F, H, and J are treated with a mouse monoclonal antibody against FGFR1 at a
concentration of 2 mg/ml. All endometrial samples shown are from patients with
no abnormal bleeding. Panels A and B show early proliferative endometrium with
moderate glandular staining and stromal cells showing significant cytoplasmic
staining. Panels C and D show a sample of day 17 endometrium. The glandular
staining is similar with no staining of the endometrial stroma. Panels E and F
show day 24 endometrium. Significant cytoplasmic staining is seen in the stroma
as well as the glandular staining. Panels G and H show day 22 endometrium with
part of the underlying myometrium. Discrete staining is seen in the glands with
minimal endometrial stromal staining. The myometrial smooth muscle cells shows
strong staining. Panels I and J show a leiomyoma with light staining throughout
the extracellular matrix and some cell associated staining but overall decreased
staining when compared to that of normal myometrium.
Figure 2. Immunohistochemical localization of
fibroblast grwoth factor receptor type 1 (FGFR1) in human uterine tissues
throughout the menstrual cycle. Panels A, C, E, G and I are negative controls
and were treated with nonspecific (Ig)G at a concentration of 5 ug/ml. Panels B,
D, F, H, and J are treated with a mouse monoclonal antibody against FGFR1 at a
concentration of 2 mg/ml. All endometrial samples shown are from patients with
no abnormal bleeding. Panels A and B show early proliferative endometrium with
moderate glandular staining and stromal cells showing significant cytoplasmic
staining. Panels C and D show a sample of day 17 endometrium. The glandular
staining is similar with no staining of the endometrial stroma. Panels E and F
show day 24 endometrium. Significant cytoplasmic staining is seen in the stroma
as well as the glandular staining. Panels G and H show day 22 endometrium with
part of the underlying myometrium. Discrete staining is seen in the glands with
minimal endometrial stromal staining. The myometrial smooth muscle cells shows
strong staining. Panels I and J show a leiomyoma with light staining throughout
the extracellular matrix and some cell associated staining but overall decreased
staining when compared to that of normal myometrium.
Utilizing reverse transcription (RT)-PCR, mRNA for
all three forms of the FGFR1: 432 bp (complete transmembrane), 165 bp
(alternatively spliced transmembrane) and 341 bp (secreted), were identified in
the endometrium, myometrium and leiomyomas (Figure 3 A, B). Additionally, the
primers designed to amplify the transmembrane forms also amplified a 540 bp
fragment of a second secreted form of FGFR1 (Figure 3A, B) (Eisemann et al.,
1991). For ease of discussion we will hereafter identify the secreted form
amplified as the 540bp fragment as secreted form A and the form detected as the
341 bp fragment as secreted form B (Eisemann et al., 1991; Johnson et al.,
1991).
There was no menstrual cycle variability seen in any tissue for any of the identified bands. Restriction digestion supported the identification of these RT-PCR products and sequencing confirmed these findings (Figure 3C, D).
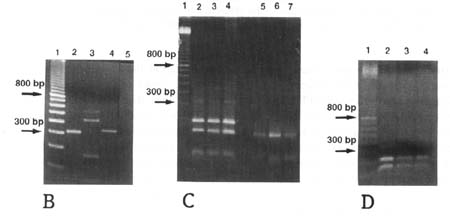
Figure 3. Reverse transcription-polymerase
chain reaction (RT-PCR) detection of fibroblast grwoth factor receptor type 1
(FGFR1) isotypes. (A) A schematic diagram of the primers used and
fragments amplified. The three extracellular immunoglobulin-like domains (Ig)
are diagrammed leading to the transmembrane portion of the molecule. Primers are
indicated with capital letters for ease of reference and fragments are
diagrammed in portion and with relative sizes represented. The sequence of
downstream primer C is repeated in both of the first two immunoglobulin domains
(Ig I and Ig II) and thus led to amplification of secreted form A in addition to
the anticipated fragments. (B) Amplification of the complete and
alternatively spliced transmembrane forms of the FGFR1 from human endometrium.
Molecular weight standards are seen in lane 1. Lane 2: a 315 bp product
representing the complete form, Lane 3: 540, 432, and 165 bp products are seen.
The 432 and 165 bp products are consistent with both the complete and
alternatively spliced transmembrane forms, respectively. The 540 bp product was
sequenced and identified as a secreted form of the receptor (form A). Lane 4
represents the 341 bp product for secreted form B. Lane 5 shows a sample that
did not receive reverse transcriptase and thus serves as a negative control.
Identical results were seen from 8 other endometrial specimens and similar
numbers of leiomyoma and myometrial samples. (C) Restriction digests for
both the complete and alternatively spliced forms of the receptor. Lane 1 is the
molecular weight standard. Lanes 2, 3 and 4 represent Ava II digestions of
amplified samples from amplification of endometrium, myometrium and leiomyoma,
respectively from a single patient. Both the 183 and the 132 bp fragment are
seen in all lanes. Lanes 5, 6 and 7 show HinfI digestion of the same samples to
identify the alternatively spliced form. The 130 bp fragment is seen in all
lanes and the 35 bp fragment is unable to be visualized due to its small size.
Identical results were seen for 7 other triplicate samples and leiomyoma/myometrium
pairs for normal women. (D) Restriction digestion of the amplified
secreted FGFR1 form B utilizing AvaII shows both the 219 and the 122 bp product
for leiomyomas (lane 2), myometrium (lane 3) and endometrium (lane 4) from a
single patient. Molecular weight markers are seen in lane 1. Identical results
were seen in tissue samples from 8 other patients and myometrial and endometrial
samples obtained from normal women.
To determine whether mRNAs for FGFR1 identified by
RT-PCR were translated into proteins, samples of human myometrium, endometrium
and leiomyomas were analyzed using western immunoblots to identify the different
protein isoforms (Figure 4). Utilizing a polyclonal FGFR1 antibody that
recognizes all forms of FGFR1 four major bands were identified in myometrium,
endometrium and in leiomyomas. The two largest proteins (167 kD and 150 kD) were
consistent with the complete and alternatively spliced transmembrane forms of
the FGFR1. Two smaller proteins (70 kD and 61 kD) are consistent with secreted
forms of the receptor (Duan et al., 1992). As expected, the molecular weights
did not change under reducing conditions.
Figure 4. Immunoblot for fibroblast grwoth factor receptor type 1 (FGFR1) isotypes. Protein molecular weight standards (st) are shown. The remaining lanes show proteins obtained from endometrium (E), leiomyomas (L) and myometrium (M) from a single patient. All four proteins were detected equally in each tissue types although due to the efficiency of protein detection each sample did not always contain all four bands. A representative gel of the five gels run is shown with all four bands can be seen for leiomyoma and three of the four bands for myometrium and endometrium.
Discussion
This study demonstrates that expression of FGFR1 in human uterine tissues shows menstrual cycle-specific regulation and differs in women with abnormal uterine bleeding and normal women. In women with abnormal uterine bleeding and leiomyomas, expression of FGFR1 in the endometrial stromal cells is dysregulated in the early luteal phase. This time coincides with the time of embryo apposition and implantation. Since the FGF system appears to play an important role in pattern formation in the early embryo, implantation and trophoblast outgrowth, this finding may have implications for women with leiomyomas who also experience infertility or recurrent miscarriage (Carlone and Rider, 1993; Cornell et al., 1995; Haimovici and Anderson, 1993).
This study also provides anatomic evidence that the bFGF receptor/ligand system may be important in the pathogenesis of leiomyoma-related bleeding. A number of studies have suggested that vascularity is abnormal in the myomatous uterus (Stewart and Nowak, 1996). Abnormalities of bFGF and FGFR1 can lead to vascular abnormalities in a number of ways that could result in abnormal bleeding. First, bFGF is an angiogenic growth factor that can induce endothelial cell proliferation thus leading to an increased number of vessels (Presta, 1988). Alternatively, bFGF regulates the production of enzymes that cause remodeling of the ECM, including collagenase and plasminogen activator which could lead to vascular dilatation and resulting in increased bleeding in this way (Presta, 1988). Finally, the bFGF receptor/ligand system may cause bleeding by disrupting the expression of integrins, cell adhesion molecules integral to the process of angiogenesis (Klein et al., 1993). bFGF had been shown to upregulate the synthesis of §3 integrin and the expression of the av§3 integrin shows the same luteal phase suppression seen in our study for FGFR1. (Lessey et al., 1992; Lessey et al., 1994).
The secreted form of the receptor identified in this study may play a unique role in the physiology or pathophysiology of the adult human uterus, particularly the leiomyomatous uterus. Although secreted forms of the FGFR1 have been identified in other adult tissues, neither of the secreted forms we report were previously identified in studies of the human fetal uterus or mature rat uterus (Hanneken et al., 1994; Hanneken and Baird, 1995; Isacchi et al., 1990; Rider et al., 1995). Previous studies from our lab demonstrated that leiomyomas contain more bFGF than normal myometrium (Mangrulkar et al., 1995). The immunostaining for FGFR1 in this study in the extracellular matrix of leiomyomas probably represents secreted receptor since there are no cell membrane to anchor the transmembrane forms. The extracellular matrix of leiomyomas may act as a reservoir of bFGF bound to secreted receptor. Thus, in addition to disordered expression in the endometrium of FGFR1, women with leiomyoma-related bleeding may also have a reservoir of bound bFGF ligand that can act in a local endocrine manner on the endometrium.
This dysregulation of the FGF receptor/ligand system may be relevant to other states characterized by abnormal uterine bleeding. For example, women using progestin-only contraceptives have abnormal uterine bleeding and their endometrium is characterized by morphologically abnormal blood vessels (Hourihan et al., 1991; Rogers et al., 1993). Further work is needed to examine the FGF system in other states associated with abnormal bleeding.
If the bFGF receptor/ligand system is important in the pathogenesis of abnormal uterine bleeding, therapeutic strategies may be developed that target these molecules. Interferons and the coupling of bFGF to cytotoxic agents including saporin are two such agents that are currently available for this purpose (David, 1995; Ezekowitz et al., 1992; Lappi and Baird, 1991; Oleszask, 1988; Real et al., 1986; White, et al., 1989). Other agents that interfere with this receptor/ligand system may also be therapeutic options for abnormal uterine bleeding.
The current study also highlights an inherent problem in many studies that utilize endometrial tissue from unspecified hysterectomy specimens as normal control samples. Better study design will lead to a better understanding of the pathophysiology of this process. By better understanding the biology of processes resulting in abnormal uterine bleeding, we may be able to design innovative therapies with greater specificity and decreased side-effects.
Acknowledgements
The authors would like to thank Dr. Virginia Rider for her expert technical advice on the cell membrane preparations and for the rat membrane extracts. This study was supported by HD-30496, National Institutes of Health, Bethesda, MD (to R.A.N.) and F32 CA-60447-03 (to B.J.Q.)
References
Budwit-Novotny, D.A., McCarty, K.S., Cox,
E.B. et al. (1986) Immunohistochemical analysis of estrogen receptor in
endometrial adenocarcinoma using a monoclonal antibody. Canc Res. 46:5419-5425.
Carlone D.L. and Rider V. (1993) Embryonic modulation of
basic fibroblast growth factor in the rat uterus. Biol. Reprod. 49:653-65.
Chirgwin, J.M., Przbyla,A.E., MacDonald, R.J., et al.
(1979) Isolation of biologicaly active ribonucleic acid from sources enriched in
ribonuclease. Biochemistry 18:5294-5300.
Cornell, R.A., Musci, T.J., and Kimelman, D. (1995) FGF is
a prospective competence factor for early activin-type signals in Xenopus
mesoderm induction. Development 121:2429-37.
David, M. (1995) Transcription factors in interferon
signaling. Pharmacol Ther. 65:149-61.
Duan, D.R., Werner, S., and Williams L. (1992) A naturally
ocurring secreted form of fibroblast growth factor receptor 1 binds basic FGF in
preference over acidic FGF. J Biol Chem.267:16076-80.
Eisemann, A., Ahn, J.A., Graziani, G., et al. (1991)
Alternative splicing generates at least five diffferent isoforms of the human
basic-FGF receptor. Oncogene 6:1195-1202. Ezekowitz, R.A., Mulliken, J.B., and
Folkman, J. (1992) Interferon alpha-2a therapy for life-threatening hemangiomas
of infancy. N Engl J Med. 326:1456-63.
Ferriani, R.A., Charnock-Jones D.S., Prentice, A., et al.
(1993) Immunohistochemical localization of acidic and basic fibroblast growth
factors in normal human endometrium and endometriosis and the detection of their
mRNA by polymerase chain reaction. Hum Reprod. 8:11-16.
Folkman J. and Klagsbrun M. (1987) Angiogenic factors.
Science, 235:442-4.
Gospodarowicz, D. (1983) Growth factors and their action
in vivo in in vitro. J. Pathol. 141:201-33.
Haimovici F. and Anderson D.J. (1993) Effects of growth
factors and growth factor-extracellular matrix interactions on mouse trophoblast
outgrowth in vitro. Biol. Reprod. 49:124-30.
Hanneken, A., Ying, W., Ling, N., et al. (1994)
Identification of soluble forms of the fibroblast growth factor receptor in
blood. Proc. Natl Acad Sci USA. 91:9170-4.
Hanneken, A. and Baird, A. (1995) Soluble forms of the
high-affinity fibroblast growth factor receptor in human vitreous fluid. Invest.
Opthalmol. Vis. Sci. 36:1192-6.
Hourihan, H.M., Sheppard, E.M., Belsey, I.A., et al.
(1991) Endometrial vascular features prior to and following exposure to
levonorgestrel. Contraception 43:375-85.
Irwin J.C., Utian, W.H., and Eckert R.L. (1991) Sex
steroids and growth factors differentially regulate the growth and
differentiation of cultured human endometrial stromal cells. Endocrinology
129:2385-92.
Isacchi, A. Bergonzoni, L., and Sarmientos, P. (1990)
Complete sequence of a human receptor for acidic and basic fibroblast growth
factors. Nucleic Acids Res 18:1906.
Johnson, D.E., Lu, J. Chen, H., et al. (1991) The human
fibroblast growth factor receptor genes: a common structural arrangement
underlies the mechanism for generating receptor forms that differ in their third
immunoglobulin domain. Mol. Cell.Biol. 9:4627-34.
Klein, S., Giancott, M., Presta, M., et al. (1993) Basic
fibroblast growth factor modulates integrin expression in microvascular
endothelial cells. Mol. Biol. Cell. 4:973-82.
Lappi, D.A. and Baird, A. (1991) Mitotoxins: growth
factors-targeted cyctotoxic molecules. Prog Growth Factor Res. 2:223-36.
Lee, P.L, Johnson, D.E., Cousens, L.S., et al. (1989)
Purification and complementary DNA cloning of a receptor for basic fibroblast
growth factor. Science 245:57-60.
Lessey, B.A., Castelbaum, A.J., Buck, C.A., et al. (1994)
Further characterization of endometrial integrins during the menstrual cycle and
in pregnancy. Fertil Steril. 62:497-506.
Lessey, B.A., Damjanovich, L., Coutifaris, C., et al.
(1992) Integrin adhesion molecules in the human endometrium. Correlation with
the normal and abnormal menstrual cycle. J Clin Invest. 90:188-95.
Mangrulkar, R.S., Ono, M., Ishikawa, M., et al. (1995)
Isolation and characterization of heparin-binding growth factors in human
leiomyomas and normal myometrium. Biol Reprod 53:636-46.
Mukko, V.R., and Stancel, G.M. (1985) Regulation of
epidermal growth factor receptor in human fetal ovary and uterus. J. Biol Chem.
260:9820-4.
Nowak, R.A., Rein, M.S., Heffner, L.G., et al. (1993)
Production of prolactin by smooth muscle cells cultured from human uterine
fibroid tumours. J Clin Endorinol Metab. 5:1308-13
Noyes, R.W., Hertig,A.T., and Rock, J. (1950) Dating the
endometrial biopsy. Fertil Steril. 1:3-25.
Oleszask, E. (1988) Inhibition of mitogenic activity of
PDGF, EGF, and FGGF by interferon-gamma. Exp Cell Res. 179:575-80.
Presta, M. (1988) Sex hormones modulate the synthesis of
basic fibroblast growth factor in human endometrial adenocarcinoma cells:
implication for the neovascularization of normal and neoplastic endometium. J
Cell Physiol. 137:593-7.
Real, F.X., Oettgen, H.F., and Krown, S.E. (1986) Kaposi's
sarcoma and the acquired immunodeficiency syndrome:treatment with high and low
doses of recombinant leukocyte A interferon. J Clin Oncol. 4:544-51.
Rider, V., Piva, M., Cohen, M., et al. (1995) Alternative
splicing and differential targeting of fibroblast growth factor receptor 1 in
the pregnant rat uterus. Endocrinology 136:3137-45.
Rogers, P.A., Au, C.L., and Affandi, B. (1993) Endometrial
microvascular density during the normal menstrual cycle and following exposure
to long-term levonorgestrel. Hum Reprod. 8:1396-1404.
Rusnati, M., Casarotti, G., Pecorelli, S., et al. (1990)
Basic fibroblast growth factor in ovulatory cycle and postmenopausal human
endometrium. Growth Factors 3:299-307.
Salat-Baroux J., Romain S., Alvarez S., et al. (1994)
Biochemical and immunohistochemical multiparametric analysis of steroid
receptors and growth factor receptors in human normal enndometrium in
spontaneous cycles and after the induction of ovulation. Hum Reprod. 9:200-8.
Stewart, E.A. and Nowak, R.A. (1996) Leiomyoma-related
bleeding: a classic hypothesis updated for the molecular era. Hum Reprod Update
2:295-306.
White, C.W., Sondheimer, H.M., Crouch, E.C., et al. (1989)
Treatment of pulmonary hemangiomatosis with recombinant interferon alpha-2a. N
Engl J Med 320:1197-200.
Yeh, J., Osathanondh, R., and Villa-Komaroff L. (1993)
Expression of messenger ribonucleic acid for epidermal growth factor and
transforming growth factor alpha, in human first-and second trimester fetal
ovary and uterus. Am J Obstet Gynecol. 168:1569-73.
Yeh, J. and Osanthanondh R. (1993) Expression of messenger
ribonucleic acids encoding for basic fibroblast growth factor (FGF) receptor in
human fetal ovary and uterus. J Clin Endorinol Metab. 77:1367-71.